B) False
Correct Answer

verified
False
Correct Answer
verified
True/False
A chemical change alters the chemical composition of a substance,and therefore a new substance is produced.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Consider the balanced reaction: 4 NO2 + O2 + 2 H2O → 4 HNO3. If 100. g of NO2 is placed in a reaction vessel the theoretical yield of nitric acid (HNO3)collected will be 137 g.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula weight of KCl?
A) 74.55 amu
B) 66.42 amu
C) 36.00 amu
D) 1386 amu
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which chemical equation is properly balanced?
A) SO2 + O2 + H2O → H2SO4
B) 2 SO2 + O2 + 2 H2O → 2 H2SO4
C) SO2 + O2 + 4 H2O → 2 H2SO4
D) 4 SO2 + O2 + 4 H2O �→ 4 H2SO4
F) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In redox reactions,metals tend to undergo oxidation.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose the theoretical yield in a reaction is 10.5 g and the percent yield is 75.5%. What is the actual yield of product obtained?
A) 793 g
B) 7.93 g
C) 13.9 g
D) 0.139 g
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass of 3.4 × 1020 molecules of ethanol (C2H6O) expressed in milligrams?
A) 0.026 mg of ethanol
B) 26,000 mg of ethanol
C) 0.012 mg of ethanol
D) 26 mg of ethanol
F) None of the above
Correct Answer

verified
D
Correct Answer
verified
Short Answer
A chemical equation is an expression that uses chemical formulas and other symbols to illustrate what ________ constitute the starting materials in a reaction and what ________ are formed.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium metal (K) reacts violently when added to water according to the balanced equation: 2 K(s) + 2 H2O(l) → 2 KOH(aq) + H2(g) . How many moles of H2O are needed to react completely with 7.54 mol of K?
A) 2 mol of H2O
B) 7.54 mol of H2O
C) 15.1 mol of H2O
D) 3.77 mol of H2O
F) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
When the mass of a substance produced in a reaction must be calculated,first its number of moles is determined using mole ratios,and then Avogadro's number is used to convert moles to grams.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula weight of Co(NO3) 3?
A) 88.94 amu
B) 244.96 amu
C) 216.94 amu
D) 148.96 amu
E) 196.96 amu
G) C) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which reaction is a double displacement reaction?
A) 3 Mg + Fe2O3 → 2 Fe + 3 MgO
B) 2 KBr + Cl2 → 2 KCl + Br2
C) 2 H2 + O2 → 2 H2O
D) 3 Ca(OH) 2 + Al2(SO4) 3 → 3 CaSO4 + 2 Al(OH) 3
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the balanced redox reaction: 2 C2H6(g) + 7 O2(g) → 4 CO2(g) + 6 H2O(g) ,which species is oxidized?
A) C2H6(g)
B) O2(g)
C) CO2(g)
D) H2O(g)
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write a balanced chemical equation for the reaction of acetone (C3H6O) with oxygen (O2) to form carbon dioxide (CO2) and water (H2O) .
A) C3H6O + O2 → CO2 + H2O
B) C3H6O + 4 O2 → 3 CO2 + 3 H2O
C) 3 CO2 + 3 H2O → C3H6O + 4 O2
D) 6 C3H6O + 8 O2 → 6 CO2 + 6 H2O
F) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
A compound that gains electrons in a redox reaction while causing another compound to be oxidized is called a(n)________ agent.
Correct Answer

verified
Correct Answer
verified
True/False
A mole of copper atoms has more atoms than a mole of lead atoms.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The actual yield of a product in a chemical reaction should not exceed its theoretical yield.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the mixture of Cl2 and F2 in a closed container as illustrated below. What will the contents of the container look like if the molecules undergo the reaction: Cl2(g) + 3 F2(g) → 2 ClF3(g) ? 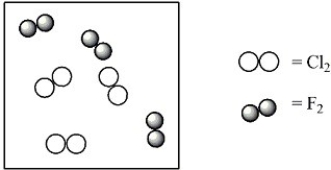
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the oxidation of sodium metal to sodium oxide described by the balanced equation: 4 Na + O2 → 2 Na2O. If 2.55 mol of sodium reacts,and 75.0 g of Na2O is produced,what is the percent yield?
A) 94.9% yield
B) 34.0% yield
C) 47.5% yield
D) 190.% yield
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 98
Related Exams